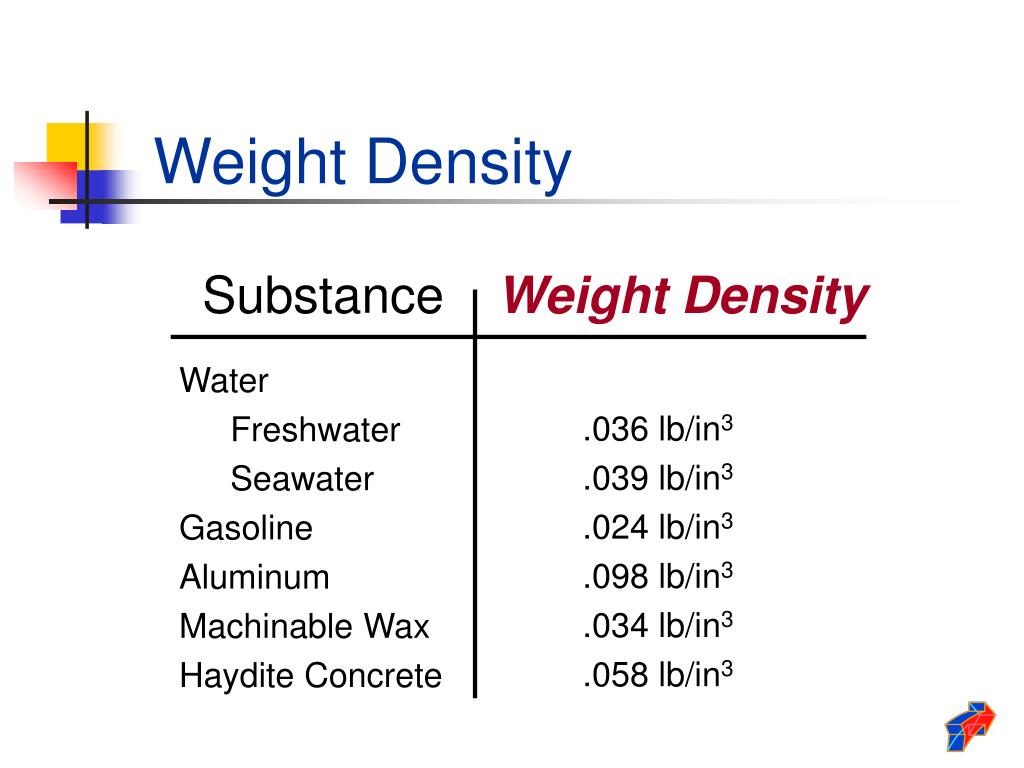
This technique can display protein molecules, organelle structures and lipid bilayers with very low degrees of distortion.Ĭondensation Clouds, formed by condensed water vapor Typically the specimens are prepared by cryofixation and freeze-fracture, after which the broken surface is freeze-etched, being eroded by exposure to vacuum till it shows the required level of detail. Sublimation is important in the preparation of certain classes of biological specimens for scanning electron microscopy. This is extremely valuable to certain scientific disciplines, a dramatic example being the collection of meteorites that are left exposed in unparalleled numbers and excellent states of preservation. As a result, there are large areas where millennial layers of snow have sublimed, leaving behind whatever non-volatile materials they had contained. Antarctica shows this effect to a unique degree because it is by far the continent with the lowest rate of precipitation on Earth. Sublimation accounts for the slow mid-winter disappearance of ice and snow at temperatures too low to cause melting. Sublimation is the process by which water molecules directly leave the surface of ice without first becoming liquid water. Įlectron micrograph of freeze-etched capillary tissue Sublimation Humidity ranges from 0 grams per cubic metre in dry air to 30 grams per cubic metre (0.03 ounce per cubic foot) when the vapor is saturated at 30 ☌. 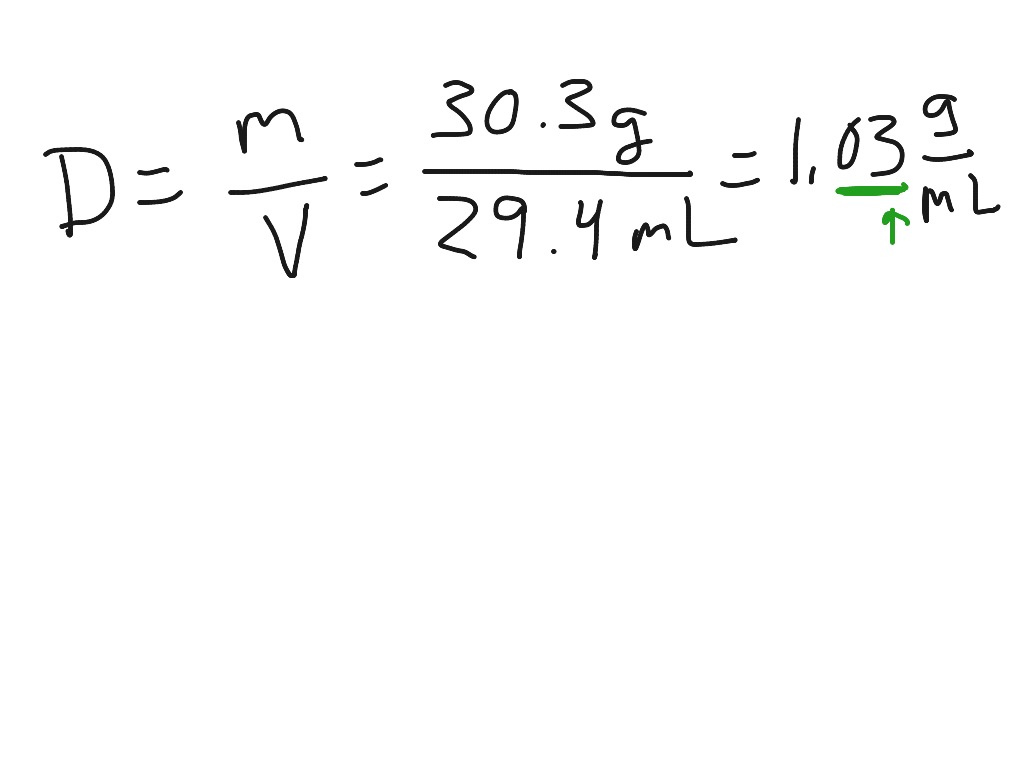
This condition is often referred to as complete saturation. The temperatures of the atmosphere and the water surface determine the equilibrium vapor pressure 100% relative humidity occurs when the partial pressure of water vapor is equal to the equilibrium vapor pressure. The measurements are usually expressed as specific humidity or percent relative humidity. The vapor content of air is measured with devices known as hygrometers. Humidity is the amount of water vapor in the air. In some countries, the evaporation rate far exceeds the precipitation rate.Įvaporative cooling is restricted by atmospheric conditions. Formulas can be used for calculating the rate of evaporation from a water surface such as a swimming pool. The measurements range from under 30 to over 120 inches per year. The US data is collected and compiled into an annual evaporation map. In the US, the National Weather Service measures the actual rate of evaporation from a standardized "pan" open water surface outdoors, at various locations nationwide. When a net evaporation occurs, the body of water will undergo a net cooling directly related to the loss of water. The amount of water vapor in the air determines how frequently molecules will return to the surface. Liquid water that becomes water vapor takes a parcel of heat with it, in a process called evaporative cooling. The aggregate measurement of this kinetic energy transfer is defined as thermal energy and occurs only when there is differential in the temperature of the water molecules. Each individual water molecule which transitions between a more associated (liquid) and a less associated (vapor/gas) state does so through the absorption or release of kinetic energy. Whenever a water molecule leaves a surface and diffuses into a surrounding gas, it is said to have evaporated. Water vapor can also be indirect evidence supporting the presence of extraterrestrial liquid water in the case of some planetary mass objects. 
Likewise the detection of extrasolar water vapor would indicate a similar distribution in other planetary systems. Water vapor is a relatively common atmospheric constituent, present even in the solar atmosphere as well as every planet in the Solar System and many astronomical objects including natural satellites, comets and even large asteroids. Use of water vapor, as steam, has been important for cooking, and as a major component in energy production and transport systems since the industrial revolution. It is less dense than most of the other constituents of air and triggers convection currents that can lead to clouds and fog.īeing a component of Earth's hydrosphere and hydrologic cycle, it is particularly abundant in Earth's atmosphere, where it acts as a greenhouse gas and warming feedback, contributing more to total greenhouse effect than non-condensable gases such as carbon dioxide and methane. Under typical atmospheric conditions, water vapor is continuously generated by evaporation and removed by condensation. Water vapor is transparent, like most constituents of the atmosphere. Water vapor can be produced from the evaporation or boiling of liquid water or from the sublimation of ice. It is one state of water within the hydrosphere. Water vapor, water vapour or aqueous vapor is the gaseous phase of water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
